 In addition to stars, plasmas are found in some other high-temperature environments (both natural and man-made), such as lightning strikes, certain television screens, and specialized analytical instruments used to detect trace amounts of metals. The presence of these charged particles imparts unique properties to plasmas that justify their classification as a state of matter distinct from gases. A plasma is a gaseous state of matter that contains appreciable numbers of electrically charged particles ( Figure 2). 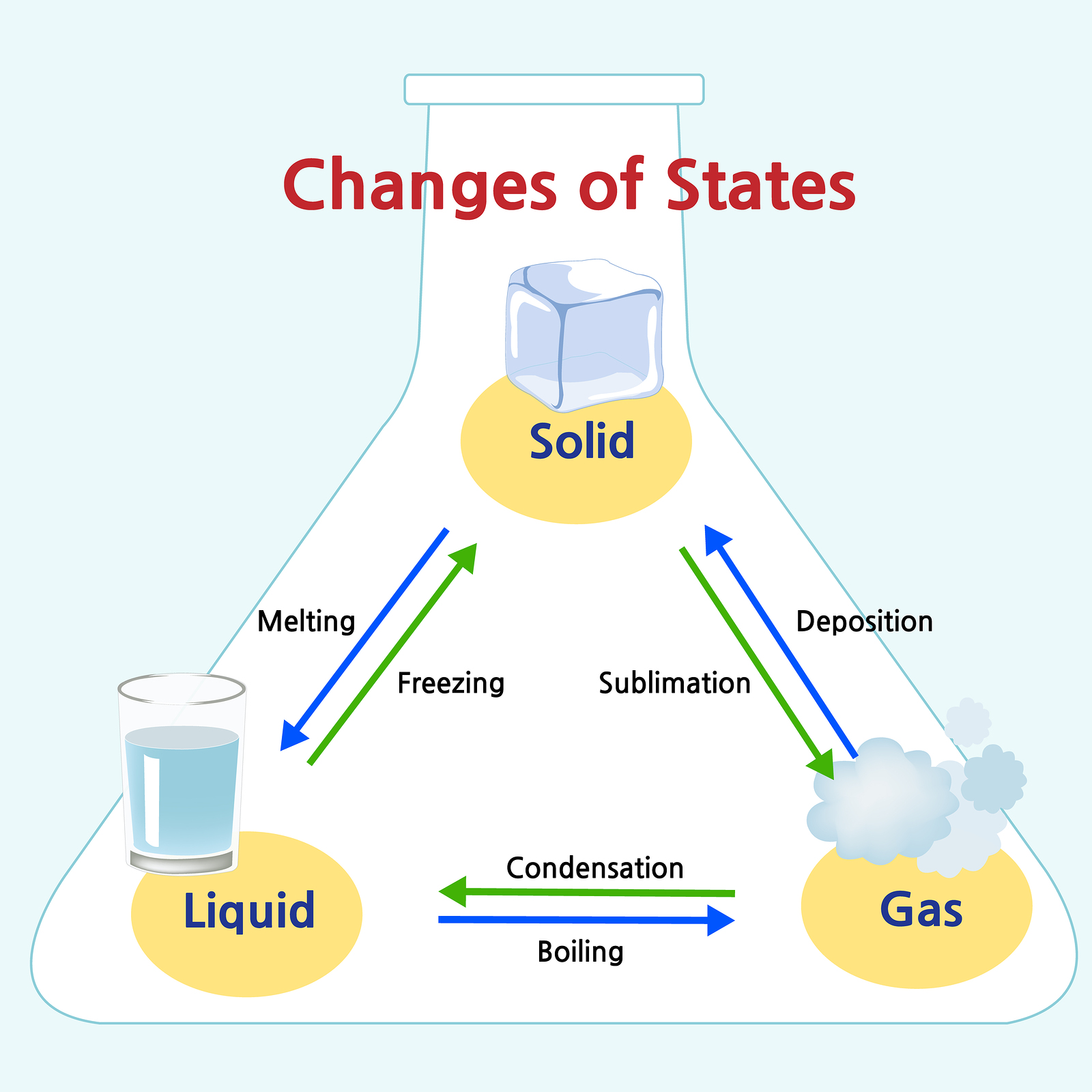
The three most common states or phases of matter are solid, liquid, and gas.Ī fourth state of matter, plasma, occurs naturally in the interiors of stars. A gas takes both the shape and volume of its container. (In zero gravity, liquids assume a spherical shape.) Both liquid and solid samples have volumes that are very nearly independent of pressure. A liquid flows and takes the shape of a container, except that it forms a flat or slightly curved upper surface when acted upon by gravity. A solid is rigid and possesses a definite shape. Solids, liquids, and gases are the three states of matter commonly found on earth ( Figure 1). Gases are also matter if gases did not take up space, a balloon would stay collapsed rather than inflate when filled with gas. 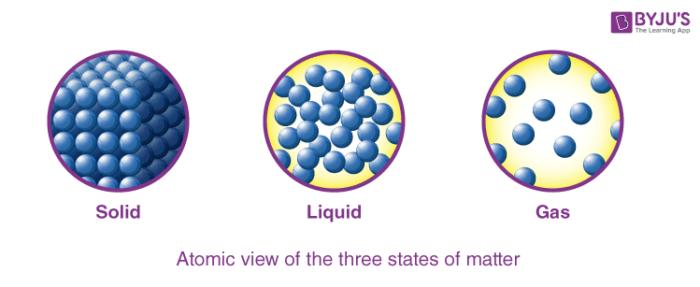
Solids and liquids are more obviously matter: we can see that they take up space, and their weight tells us that they have mass. Matter is defined as anything that occupies space and has mass, and it is all around us. | Key Concepts and Summary | Glossary | End of Section Exercises | Matter Matter on the Macroscopic Scale Identify and describe the basic properties of each physical state of matter: solid, liquid, and gas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
